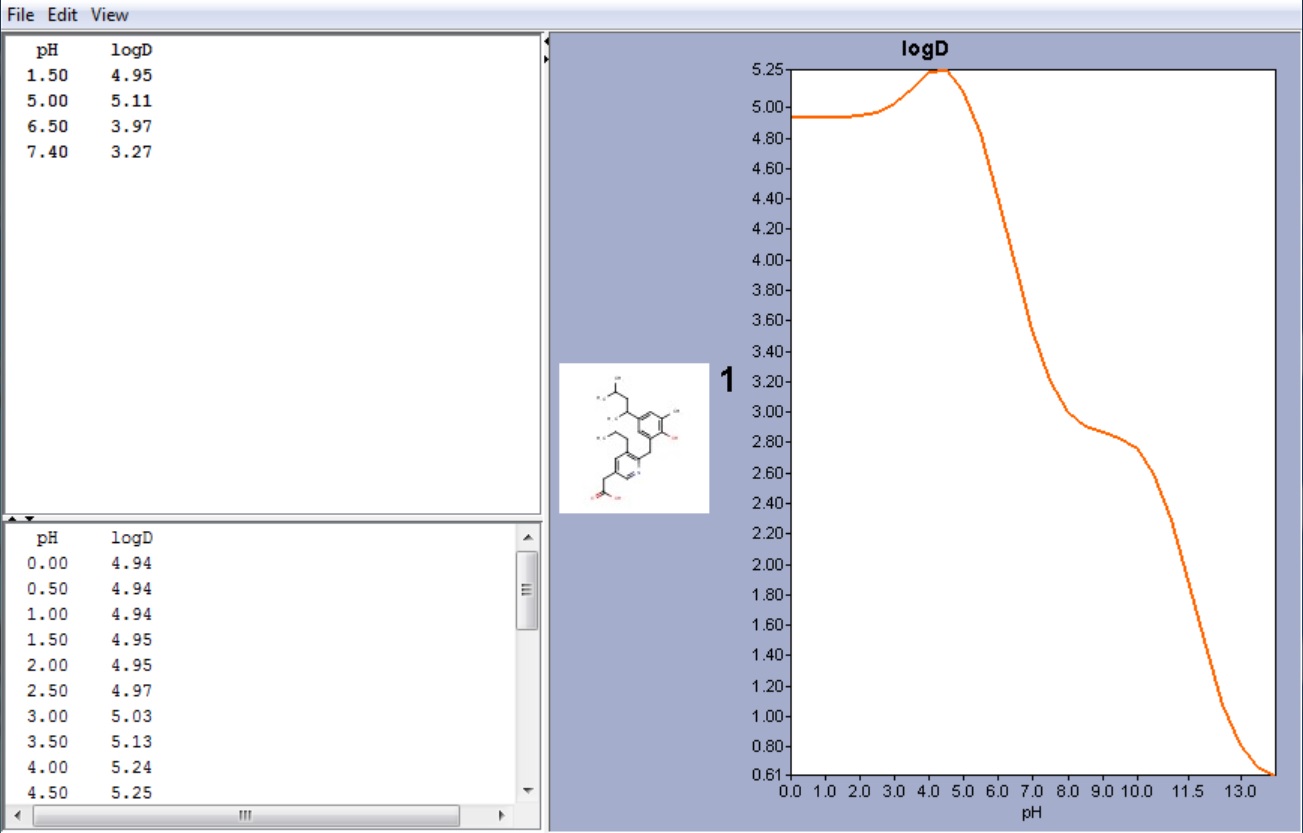
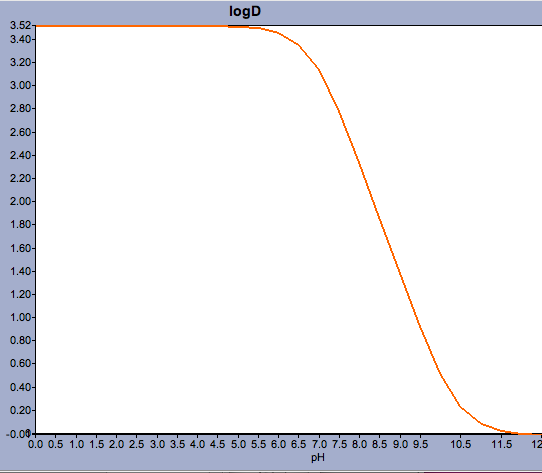
SOLVED: For an acidic drug, the following relationship between logD and logP exists when pH is much greater than pKa of the drug: LogD = LogP + (pH - pKa) Derive the

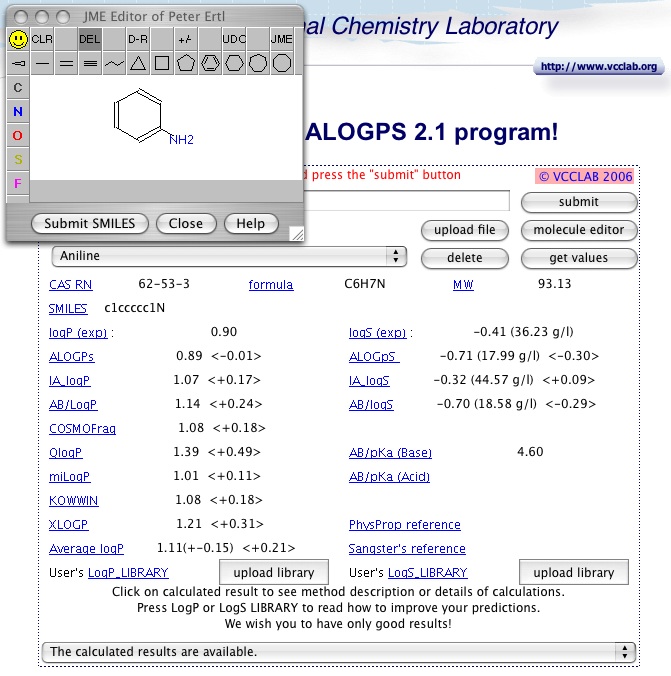
Systematic Modeling of log D7.4 Based on Ensemble Machine Learning, Group Contribution, and Matched Molecular Pair Analysis | Journal of Chemical Information and Modeling

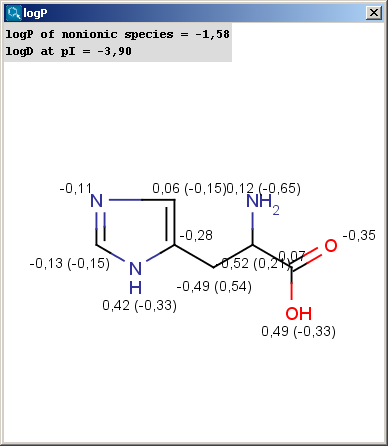
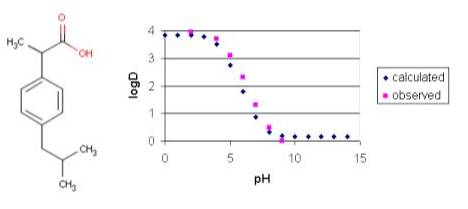
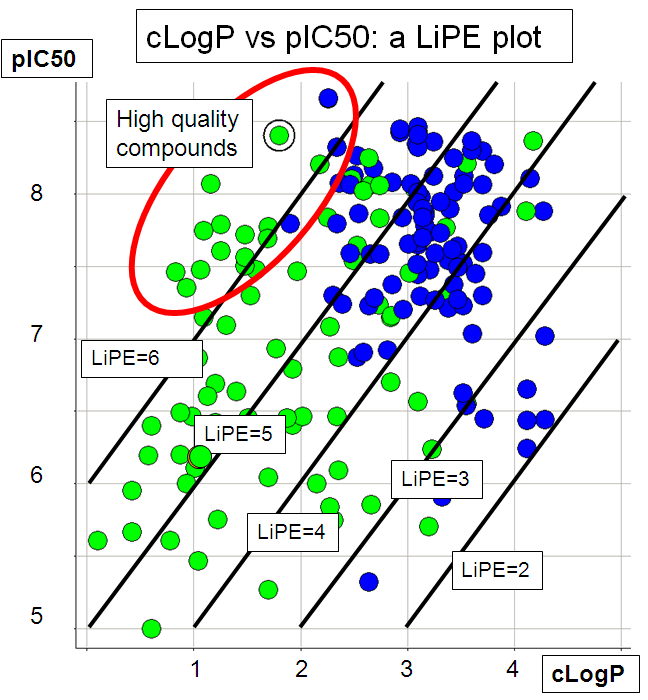











![Classification of Log P calculation methods according to Mannhold [1] | Download Table Classification of Log P calculation methods according to Mannhold [1] | Download Table](https://www.researchgate.net/publication/227111946/figure/tbl1/AS:668925599694851@1536495699250/Classification-of-Log-P-calculation-methods-according-to-Mannhold-1.png)